ADAPT: The Platform Redefining Drug Discovery
A unified, AI-driven ecosystem that removes the bottlenecks of traditional R&D.
What ADAPT Is
ADAPT (Advanced Drug Discovery Automation Platform) is a modular platform that integrates semantic mining, structural modeling, predictive chemistry, and automated experimentation into a single accelerated workflow.
It streamlines the entire path from literature mining to preclinical validation, reducing time and cost while increasing precision and scalability.
HOW ADAPT WORKS
Generative and predictive models propose new molecules and materials.
Robotic systems execute synthesis and assays.
Experimental results update the models in real time.
Instead of advancing through disconnected stations, the process runs in parallel and continuously self-optimizes.
CAPABILITIES
Ultra-scale virtual screening: evaluates >10¹⁴ molecules efficiently.
Multi-objective optimization: balances potency, selectivity, ADMET, synthetic feasibility, and cost.
Generative AI for materials: designs novel biomaterials with target properties.
Network inference: reconstructs interaction networks from large-scale expression data.
Next-generation multi-omics: robust cross-omics integration through a state-of-the-art international framework.
WHAT ADAPT DELIVERS
ADAPT unifies AI-driven target prediction, molecular screening, quantum-level descriptors, and synthesis planning into a single coherent pipeline.
It bridges literature mining, structure-based design, and automated validation to deliver preclinical candidates faster, more efficiently, and with higher precision.
Key Challenges & ADAPT’s Solutions
| Challenge | Traditional Limitation | ADAPT’s Innovation |
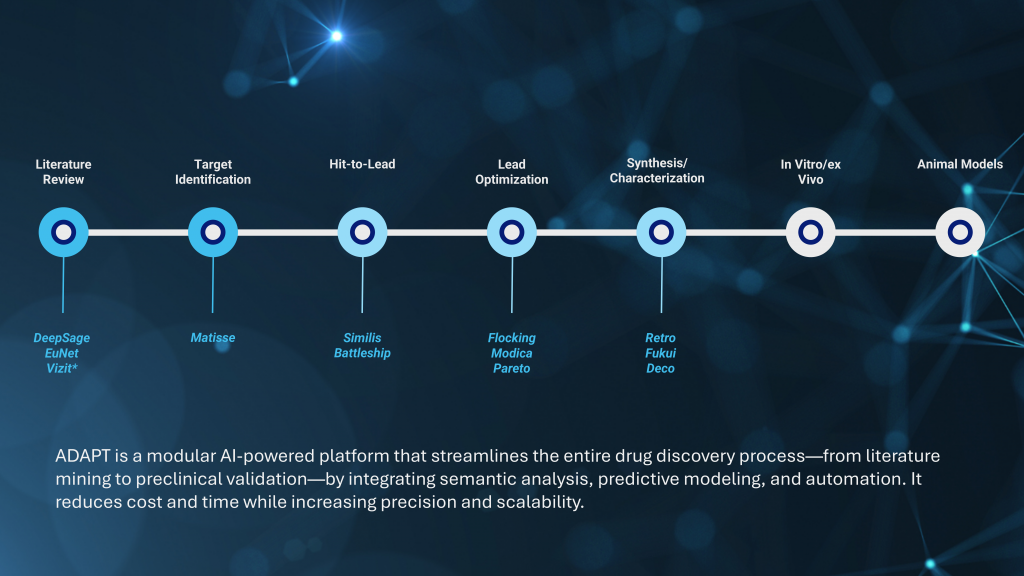
| Literature Overload | Manual review of thousands of papers is slow and error-prone | DeepSage and EuNet, along with Vizit —an external solution developed by Biovista Ltd- extract mechanistic relationships from biomedical texts using AI-based knowledge graphs |
| Target Identification | Low-resolution models and indirect biological inference | Matisse leverages 3D geometry and topological analysis of binding sites to improve target selection |
| Hit Validation | High false-positive rate, poor bioavailability | Similis and Battleship perform similarity-based screening against validated compound libraries |
| Lead Optimization | Inefficient exploration of chemical space, limited feedback | Flocking, Modica, and Pareto apply AI to optimize potency, ADMET, and synthetic accessibility in parallel |
| Synthesis Planning | Trial-and-error synthesis, costly failures | Retro, Fukui, and Deco predict reactivity and plan synthetic routes based on quantum descriptors and retrosynthetic logic |
| Data Fragmentation | Silos between in silico and experimental labs | ADAPT connects to robotic platforms (e.g., MOSQUITO SPT) and databases (e.g., PDB, ChEMBL) for seamless validation and iteration |
PARADIGM SHIFT
ADAPT replaces fragmented, hypothesis-limited workflows with an integrated, AI-augmented pipeline.
It enables researchers to move from literature to validated preclinical candidates with unprecedented speed, traceability, and precision.
How to cite us
A semantic literature analysis was performed using DeepSage (ADAPT platform, SoftMining, adapt.softmining.it), an AI-powered tool designed to extract and organize scientific knowledge from large-scale databases. DeepSage facilitated the identification of relevant research trends and conceptual frameworks, supporting the development of this study.
